Cryptomycota and Rozella

Molecular sequencing of environmental samples has brought about an awareness of unseen and previously unrealized biodiversity, labeled in part "dark matter fungi" (Grossart et al. 2016). Among these widely dispersed but poorly known organisms is a recently recognized lineage of unwalled endoparasites, the superphylum Opisthosporidia. One phylum in this superphylum is Cryptomycota.
The genus Rozella is an unwalled endoparasite of Oomycetes, Chytridiomycota, Blastocladiomycota and green algae. Because Rozella reproduces with posteriorly, uniflagellate zoospores, historically the genus has been included among the zoosporic fungi, Chytridiomycota (Barr 1980). Unexpectantly, molecular sequence analyses revealed that Rozella was sister to zoosporic fungi (James et al. 2006) and in a clade with a large number of environmental sequences (Lara et al. 2010). The phylum Cryptomycota (Jones et al. 2011) [=Rozellomycota) was based on these environmental sequences and two organisms of known morphology, Rozella allomycis and Rozella ex Rhizoclosmatium (James et al. 2006).
The photomicrograph at the left shows host hyphae of Allomyces macrogynus infected with the endoparasite Rozella allomycis. Hyphae of A. macrogynus bear their own terminal, tan sporangia. The parasite induces host hyphae to produce septated compartments. The hyphal compartments contain, dark, round resting spores and unwalled sporangia of R. allomycis. After the parasite discharges its zoospores, it leaves empty compartments.
The morphologically diversity of organisms in this group, or related to this group, is beginning to build, with morphologically characterized organisms finding affinities with clades that contain solely environmental phylotypes (Grossart et al. 2016). As listed below, additional related, unwalled endoparasites have been discovered recently, and these share characteristics with Cryptomycota as well as the phylum Microsporidia, forming the basis for the potential unification of Cryptomycota with Microsporidia as one group (Grossart et al. 2016). However, because of the rapid evolution of the genomes of these obligate parasites and the need for greater sampling, a robust phylogeny is still needed to confirm their phylogenetic and nomenclatural placement.
There are now 27 species of Rozella described, but most are poorly known or have been reported only once. They are found as endoparasites on a variety of zoosporic fungi (Blastocladiomycota, Chytridiomycota) and oomycetes, and one has been reported on algae. Species have been based primarily on host, with other characters, such as resting spore structure and impact on host morphology, as additional characters. They share the characteristics of:
The genus Rozella is an unwalled endoparasite of Oomycetes, Chytridiomycota, Blastocladiomycota and green algae. Because Rozella reproduces with posteriorly, uniflagellate zoospores, historically the genus has been included among the zoosporic fungi, Chytridiomycota (Barr 1980). Unexpectantly, molecular sequence analyses revealed that Rozella was sister to zoosporic fungi (James et al. 2006) and in a clade with a large number of environmental sequences (Lara et al. 2010). The phylum Cryptomycota (Jones et al. 2011) [=Rozellomycota) was based on these environmental sequences and two organisms of known morphology, Rozella allomycis and Rozella ex Rhizoclosmatium (James et al. 2006).
The photomicrograph at the left shows host hyphae of Allomyces macrogynus infected with the endoparasite Rozella allomycis. Hyphae of A. macrogynus bear their own terminal, tan sporangia. The parasite induces host hyphae to produce septated compartments. The hyphal compartments contain, dark, round resting spores and unwalled sporangia of R. allomycis. After the parasite discharges its zoospores, it leaves empty compartments.
The morphologically diversity of organisms in this group, or related to this group, is beginning to build, with morphologically characterized organisms finding affinities with clades that contain solely environmental phylotypes (Grossart et al. 2016). As listed below, additional related, unwalled endoparasites have been discovered recently, and these share characteristics with Cryptomycota as well as the phylum Microsporidia, forming the basis for the potential unification of Cryptomycota with Microsporidia as one group (Grossart et al. 2016). However, because of the rapid evolution of the genomes of these obligate parasites and the need for greater sampling, a robust phylogeny is still needed to confirm their phylogenetic and nomenclatural placement.
- Nucleophaga amoebae and N. terricolae (Corsaro et al. 2014b, 2016) – endoparasites in nuclei of amoebae.
- Paramicrosporidium species. (Corsaro et al. 2014a) – endoparasite in nuclei in amoebae.
- Mitosporidium daphnia (Haag et al. 2014) – endoparasite in the hind gut epithelium of water fleas (Daphnia magna).
There are now 27 species of Rozella described, but most are poorly known or have been reported only once. They are found as endoparasites on a variety of zoosporic fungi (Blastocladiomycota, Chytridiomycota) and oomycetes, and one has been reported on algae. Species have been based primarily on host, with other characters, such as resting spore structure and impact on host morphology, as additional characters. They share the characteristics of:
- zoospores with a posteriorly directed flagellum,
- an unwalled thallus, which replaces host cytoplasm and uses the host wall as its own when the thallus converts into a sporangium.
- resting spores, which are spherical, smooth-walled or spiny, laying loosely in the host.
Life history of Rozella
An unwalled, posteriorly uniflagellate motile cell attaches to a host and encysts. The encysted zoospore then penetrates the host cell wall with a short tube and discharges its protoplast into the host. The unwalled parasitic thallus enlarges, engulphing host cytoplasm, and undergoes repeated nuclear divisions. Eventually the multinucleate plasmodium cleaves into uninucleate zoospores, which are released and continue the life history. Some unwalled portions of the plasmodium round-up and form a multi-layered wall and become resting spores. There may be a single resting spore or multiple resting spores within a single host compartment or cell.
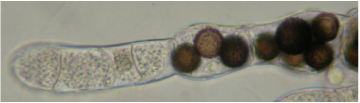
In the photomicrograph below, Allomyces macrogynus is infected with Rozella allomycis. Unwalled sporangia at the left fill host compartments. Dark-spiny walled resting spores fill host compartments at the right.
In the photomicrograph below, Allomyces macrogynus is infected with Rozella allomycis. Unwalled sporangia at the left fill host compartments. Dark-spiny walled resting spores fill host compartments at the right.
References
Barr. 1980. Canadian Journal of Botany 58: 2380-2394.
Corsaro et al. 2014a. Parasitology Research 113: 1909-1918.
Corsaro et al. 2014b. Parasitology Research 113: 4491-4498.
Corsaro et al. 2016. Parasitology Research doi:10.1007/s00436-016-5055-9
Gleason et al. 2012. Fungal Ecology 5: 651-656.
Grossart et al. 2016. Fungal Ecology 9: 28-38.
Haag et al. 2014. PNAS 111: 15480-15485
James et al. 2006. Mycologia 98: 860-871.
Jones et al. 2011. IMA Fungus 2: 173-175.
Lara et al. 2010. Protist 161: 116-121.
Corsaro et al. 2014a. Parasitology Research 113: 1909-1918.
Corsaro et al. 2014b. Parasitology Research 113: 4491-4498.
Corsaro et al. 2016. Parasitology Research doi:10.1007/s00436-016-5055-9
Gleason et al. 2012. Fungal Ecology 5: 651-656.
Grossart et al. 2016. Fungal Ecology 9: 28-38.
Haag et al. 2014. PNAS 111: 15480-15485
James et al. 2006. Mycologia 98: 860-871.
Jones et al. 2011. IMA Fungus 2: 173-175.
Lara et al. 2010. Protist 161: 116-121.